By: E.K. Koh, Chief Product Officer
Patients haven’t always been at the center of clinical trials. While patient-centricity has been a topic of conversation for a while, there hasn’t been any widespread consensus on what constitutes patient-centricity or a strong push for the industry to move in that direction. Until the pandemic. As the world shut down and went inside, study teams quickly adopted the decentralized clinical trial (DCT) model or virtual elements to continue treatment and trial progress. And the slow-burgeoning notion of decentralizing trials was suddenly moved to the top of the priority list.
It should come as no surprise that decentralized and hybrid trials are growing at an accelerated rate. According to a report from Global Data1, 673 trials in 2020 were initiated with virtual components. This number jumped to 1,011 in 2021 and it is anticipated that 2022 will have approximately 1,300 trials with decentralized and/or virtual components. This is a 93% increase in just two years.
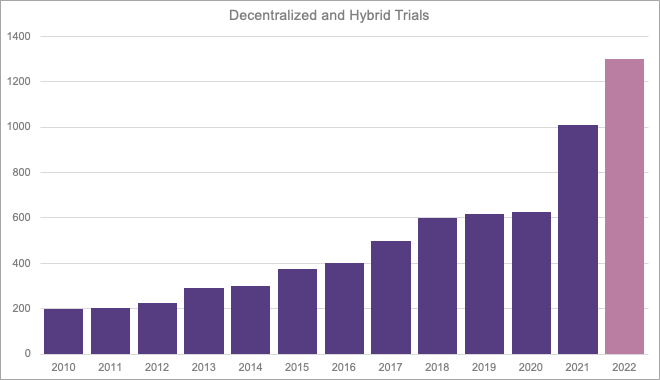
Adapted from Global Data report identifying trials with decentralized and/or virtual components in the protocol; Global Data; Virtual Clinical Trials - Thematic Research; August 21, 2021.
Using the decentralized or hybrid approach with virtual elements strongly supports a patient-centric model. Patient-centric trials are generally understood as studies that place particular emphasis on patients' perspectives and needs within the trial design and conduct.
As most trial professionals are aware, participating in a traditional clinical trial can be challenging for patients. Sites are not always conveniently located and travel to and from can be time-consuming and costly. Additionally, many patients have health risks that make participation difficult. When trials leverage virtual, decentralized elements that reach patients where they are, they remove this location barrier, enabling patients to take part and stay engaged in the trial. One study2 of decentralized vs. conventional models confirmed higher retention rates with 89% of enrolled patients in the decentralized study completing the study compared to 60% in the conventional arm.
Decentralized and hybrid elements supporting patient-centricity
Technology has played a significant role in the evolution of clinical trials overall. Existing technologies that were once considered for use in the distant future are now being fast-tracked for immediate use. Additionally, sponsors, CROs, and study teams are using systems in new and innovative ways to achieve their patient-specific goals. Consider what the following three technologies have to offer the patient experience.
eConsent. The digitization of the consent process has been an enabler for the industry. A low-risk, high-reward proposition, trial professionals are realizing the potential for improved patient comprehension and retention, as well as overall efficiencies and reduced regulatory risk and audit findings. Despite the growth in adoption, there remains some hesitancy to adopt this resource owing to perceived complexities in global trials. However, flexible eConsent systems can now accommodate both digital and paper-based consenting, which eases workloads and provides improved visibility into the consent process.
IRT. Interactive Response Technology (IRT), which manages the drug and patient logistics of clinical trials, plays a pivotal role in decentralized and hybrid models. At a high level, IRT has a primary job to fulfill - getting the right drug to the right patient at the right time. With an adequately flexible design, the IRT can support complex protocol needs at any trial stage, as well as manage the logistical complexities of remote or hybrid visit schedules for each patient. As patients have unique site visit needs, flexible visit structures and self-service capabilities make accommodating these needs easier. IRT will also manage multiple direct-to-patient shipping scenarios within a study to further support patient flexibility.
eCOA. Data collection is critical to trial progress and, ultimately, its success. Electronic Clinical Outcome Assessments (eCOAs) enable patients, caregivers, and clinicians to directly document outcomes using a variety of devices. Patients can complete assessments whenever necessary and study teams can access complete, accurate, and protected outcomes data anytime. This flexibility and convenience of the eCOA system can help reinforce a positive patient experience.
One system for better patient experience and workflow
While eConsent, IRT, and eCOA bring a great deal of value in the support of patient-oriented, decentralized trials, it’s a workflow challenge when each system operates individually. Integrations can be employed to enable data exchange, however, the staff and patient experience in this scenario are less than optimal. Not only must users log into each system individually, but they also need to thoroughly understand three disparate systems. Study teams are forced to toggle between screens to accomplish tasks, interrupting what should be a seamless workflow.
Imagine, however, logging into a single system that begins and ends with the patient - from consent to data submission. It begins with the patient logging in to give consent. Once consented, they are then screened for eligibility by staff and, when eligible, randomized into a treatment arm. In a decentralized setting, the patient’s experience continues with a home visit where study testing and procedures are performed. Direct-to-patient shipping enables the drug or device to be delivered to the patient. To report health outcomes, the patient, caregiver, or clinician simply logs into the same system as before, providing study teams with quick visibility when needed.
A unified, patient-centric platform where eConsent, IRT, and eCOA work together without interruption is an important consideration for creating an experience that fits the needs of the trial and the patient. In contrast to bundled technologies that can be disjointed, a single platform delivers the three solutions as an efficient workflow rather than as programs. As clinical trials grow more complex, using modern technological best practices in the platform ensures faster deployment of a flexible, customized system that fits the nuances of patient and trial needs while supporting ongoing compliance with regulatory agencies.
Ultimately, a patient-centric approach that leverages decentralized elements addresses the needs of patients that can be unmet in traditional settings. At the onset of any trial, taking a holistic look at the patient journey and experience during the trial design will go a long way toward helping patients feel more positively about the study and clinical trials overall.
REFERENCES
1 Figures adapted from Global Data report identifying trials with decentralized and/or virtual components in the protocol; Virtual Clinical Trials – Thematic Research; August 21, 2021.
2 Sommer C, Zuccolin D, Arnera V, Schmitz N, Adolfsson P, Colombo N, Gilg R, McDowell B. Building clinical trials around patients: Evaluation and comparison of decentralized and conventional site models in patients with low back pain. Contemp Clin Trials Commun. 2018 Jun 28;11:120-126. doi: 10.1016/j.conctc.2018.06.008. PMID: 30094387; PMCID: PMC6072894.
Author

E.K. Koh
Chief Product Officer
Suvoda
