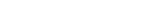RTSM (IRT) that takes weeks off study startup
Accelerate your study from contract to go-live–with agentic AI built for the most complex randomization and trial supply management.
Study startup
Up to 80% reduction in kickoff-to-UAT timelines*
*Based on preliminary testing.Fastest delivery
kickoff to UAT with agentic RTSM
On time delivery
for customer acceptance
Suvoda RTSM (IRT) supports sponsors and CROs
Start up studies faster |
Handle any protocol, quickly |
Give sponsors and sites their time back |
Stay confident in the data |
|
AI agents that cut kickoff-to-UAT timelines by up to 80%—so the path to first-patient-in is shorter. |
Purpose-built for the most complex trials—oncology, CNS, rare disease—with the speed and flexibility to adapt when requirements change. |
Sofia AI assistant. Faster access to trial information. Less administrative work. More room for the judgment calls that matter. |
Real-time visibility across subjects, sites, drugs, and depots—so every decision is grounded in what's actually happening in your study. |
The leading RTSM (IRT) that handles even the most complex protocols.
Clinical trials have never been simple. But getting a study off the ground—from contract to a working system—has always taken longer than it should. Configuration, manual testing, change orders, back-and-forth—all pull time away from what matters most: time with patients.
Suvoda RTSM changes that. It uses agentic AI to do the heavy lifting in study setup—so your team can focus on the trial, faster. Studies that once took months to set up can now be ready for User Acceptance Testing in as little as two weeks.
At its core, it does what Suvoda IRT has always done: gets the right drug to the right patient at the right time, across even the most complex protocols in oncology, CNS, and rare disease. Regardless of whether that’s complexity of the therapeutic area, operations, geography, or languages. What's new is the speed.
Suvoda RTSM—also referred to as IRT, or (Interactive Response Technology)—gives sponsors and study teams:
- Faster study startup—agentic AI that cuts kickoff-to-UAT by up to 80%
- A better experience for sponsors and sites—quicker access to information, with less friction throughout the study
- Intelligent insights—real-time visibility into trial status and the decision-support to act on it
- Complete IRT/RTSM functionality—built for complexity, adaptable when protocols change, and ready for the hardest trials
Throughout it all, Sofia, Suvoda's AI assistant, is embedded in the system. Ask it a question, pull a report, check depot inventory, or get a drug lot release in seconds. What used to take multiple screens and minutes of clicking is now a conversation.
Built for complexity. Accelerated by AI.
In Suvoda RTSM, multiple AI agents work in parallel to handle distinct phases of the study build—software configuration, customization, testing—and to turn around change orders mid-study. Because it's built on Suvoda's patented platform, it draws on what we've learned delivering thousands of trials. It doesn't start from scratch for each build. It starts from experience.
The result is a modular architecture that lets our services teams launch fast and adapt faster. When a protocol changes—and it will—mid-study amendments are a controlled, speedy process, not a project themselves.
Trial logistics
-
Agentic AI study build and configuration
-
Roles, permissions, and blinding management
-
Study and site administration
-
Cohort/stage/phase management
-
Dynamic cohort and dose management
-
Additional and dynamic visit schedule
-
Embedded, intuitive Sofia AI assistant
Patient logistics
-
Subject management
-
Adaptive replacement and randomization
-
Cross-over and re-treatment
-
Open-label extension
-
Dose calculation
-
Subject roll-over
-
Dose modification and interruption
Drug logistics
-
Drug dispensing management
-
Drug supply management
-
Drug accountability, reconciliation, and returns/destruction management
-
Temperature excursion management
-
Controlled substance management
-
Variable drug sourcing
-
Central pharmacy
-
Direct-to-patient shipping
-
Robust supply strategy management
-
Fast access to drug supply data and visualizations with Sofia AI assistant
-
Drug supply forecasting
Built to embrace the unknown. Because your RTSM should be as ready for change as you are.
The scientific process isn't static. And neither is your trial. Protocols get amended. Doses change. Sites are added. That's why Suvoda RTSM is designed to adapt mid-study.
Permissioned users can make commonly-needed modifications and corrections directly in the system after go-live. No change order, no waiting. And when more significant updates are required, agentic AI can handle the configuration work, so what once stretched across weeks can now be done in a fraction of the time.
Simple enough to use on day one. Powerful enough for your most complex protocols.
A well-designed RTSM/IRT system gets out of the way. It lets site coordinators, study teams, and even home caregivers focus on patients rather thanon navigating the software.
Suvoda RTSM is built with that in mind. Minimalist design, intuitive navigation, patient-centric workflows. Whether someone uses it every day or once a month, the experience is consistent and clear.
Sofia makes it faster still. Ask a question, pull trial data, compare depot inventories, review drug lot releases, create a graph—all in a conversation, without leaving the screen you're on. The study blind is protected. The answers are accurate. And what used to take several clicks now takes seconds.
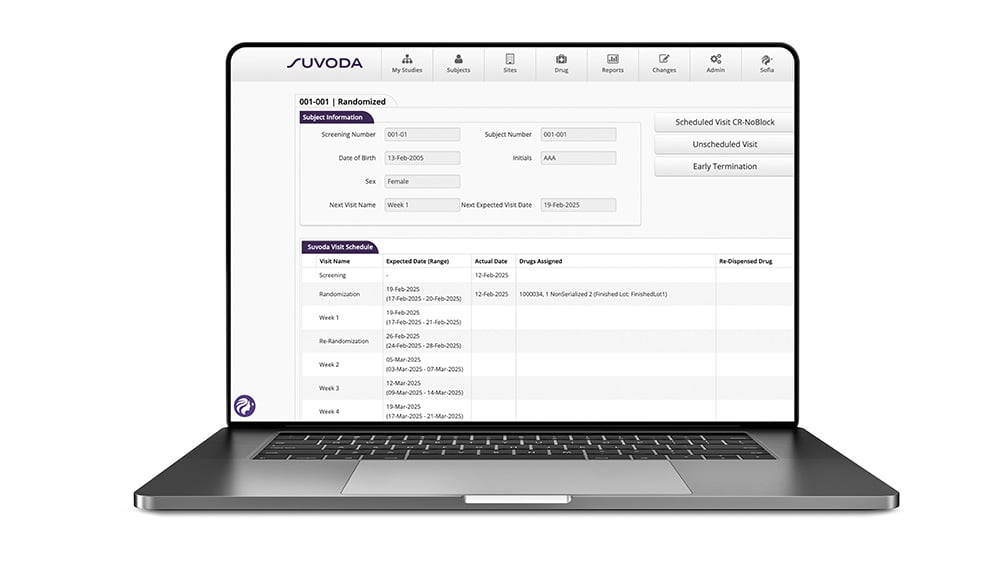
Always know where you stand—and what to do about it.
Pre-set and ad-hoc reports in Suvoda RTSM give you real-time views of the data points, KPIs, and trends that matter most: subjects, sites, drugs, depots. What you need to manage today's trial—and plan tomorrow's.
With Sofia, those insights are even more accessible. Ask for a depot inventory summary, flag a supply risk, or pull a cross-site compliance view—in the interface, in seconds, without building a report from scratch.
FEATURED IRT MODULES
RTSM FAQs
Randomization and Trial Supply Management (RTSM) is the system clinical trials use to manage patient randomization, drug assignment, and supply chain logistics—automating the workflows that keep the right drug reaching the right patient at the right time.
RTSM handles everything from patient enrollment and treatment allocation to inventory tracking, resupply, and compliance monitoring. Today, RTSM systems like Suvoda RTSM also deliver full Interactive Response Technology (IRT) capabilities, including the web-based interfaces and real-time interactions that site staff and study teams rely on throughout a trial.
IRT (Interactive Response Technology) and RTSM (Randomization and Trial Supply Management) refer to the same category of clinical trial software. The terms evolved separately —IRT describing how the system works (users interact with it in real time), RTSM describing what it does (manages randomization and drug supply)—but today they're used interchangeably across the industry.
Modern RTSM systems, including Suvoda RTSM, deliver all the capabilities that IRT has always provided, and more. If your team uses IRT, RTSM, or both, you're talking about the same system.
Agentic RTSM is Suvoda's next-generation approach to randomization and trial supply management—one that uses multiple AI agents to automate the phases of a study build that have historically been the slowest: software configuration, customization, testing, and change orders.
Because it's built on Suvoda's patented platform— it leverages the virtual partition architecture and the code generation capabilities of the platform’s low-code/no-code tools, rather than rebuilding from scratch for each build]. Studies that once took months to set up can now be ready for UAT in as little as two weeks.
Three things set Suvoda RTSM apart: agentic AI that dramatically accelerates study startup, deep expertise in complex trials, and a unified platform built on a single data model.
- Agentic AI + Sofia: Multiple AI agents automate study builds and change orders. Sofia gives users conversational access to trial data and decision support throughout the study.
- Unified platform: Single data model across RTSM, eCOA, eConsent, Patient Payments, Patient Travel, Scheduling, and the Greenphire Clinical Finance suite.
- Complex study expertise: Specialized in oncology, CNS, and rare disease. Modular, patented architecture for rapid deployment and mid-study modifications.
Powering the patient's digital journey in life-changing clinical trials

Take command of the cold supply chain
Take command of the cold supply chain
Gain control in a world of decentralized trials
Gain control in a world of decentralized trials
Broad perspective. Keen insight. Calm guidance.
Broad perspective. Keen insight. Calm guidance.
Seamless technology in complex ecosystems
Seamless technology in complex ecosystems
INSIGHTS & NEWS
-
BLOG
From buzzword to backbone: How AI is reshaping clinical trial operations
May 28, 2026 -
BLOG
Selecting the right site payments solution for today’s clinical trials: what to look for
May 21, 2026 -
ON-DEMAND
Suvoda IRT Demo
May 20, 2026 -
BLOG
Study startup has enough demands on your attention. RTSM build shouldn't be one of them
May 15, 2026 -
NEWS
Suvoda Patient App Earns “Best Patient Relationship Management Solution” In 2026 MedTech Breakthrough Awards Program
May 7, 2026 -
BLOG
A new regulatory milestone: what the joint FDA and EMA’s AI principles can mean for clinical trial technology
Apr 30, 2026 -
NEWS
Suvoda announces agentic RTSM, enabling the reduction of clinical trial startup timelines by up to 80%
Apr 21, 2026 -
BLOG
IRT vs. RTSM: what’s the difference? What sponsors need to know when choosing clinical trial technology
Apr 16, 2026 -
ON-DEMAND
Demo - eCOA unified with IRT on the Suvoda Platform
Mar 27, 2026 -
BLOG
Technology fragmentation is one of the biggest hidden risks in clinical trials
Mar 26, 2026